Boltzmann Constant and Avogadro's Number
The ideal gas constant is the combination of Boyles law Avogadros number Charless law and Gay-Lussacs law. One mol contains exactly 60221407610²³ elementary particles this number is called the Avogadros Number.

If The Value Of Avogadro Numberis 6 023xx10 23 Mol 1 And The Vaueof Boltzmann Constant Is Youtube
Because of the relatively recent definition change use care when comparing calculations prior to 2019 because the values for R are slightly different before and after the.

. Although their values are identical they describe. The molar gas constant R is defined as Avogadros number times the Boltzmann constant. As a consequence the gas constant also now has an exact value.
Electron mass energy J m e c 2. The speed probability density functions for the speeds of some noble gases calculated at a constant temperature of 29815K The distribution was first derived by Maxwell in the 1860s and later by Boltzmann in the 1870s by carrying out significant investigations into the physical origins of this distribution. The universal gas constant R is defined as the product of Avogadro constant N A number of particles in one mole of gas and Boltzmanns constant k it relates the kinetic energy of particles in a gas.
The units may be electrons ions atoms or molecules depending on the character of the reaction and the nature of the substance. The name Avogadros number was coined in 1909 by the physicist Jean Perrin who defined it as the. 8187 x 10-14 J.
Gas Constant In Different. Electron rest mass. 2818 x 10-15 m.
The digits inside the parentheses are the uncertainty in the measurement of the gas constant value. The ideal gas constant and the Boltzmann constant k B are related by Avogadros constant N A. A mole is a unit used for measuring matter.
Permittivity of vacuum 0 885 10 12 Fm. Its also the number of molecules in a mole a unit of measurement. Gravitation constant G 66710 11 m 3 kg 1 s 2.
Boltzmanns Constant k b 1381 10-23 J K-1. Apart from the above equations the gas constant is also found in many. 831446261815324 JK 1 mol 1.
Molar gas constant R 8314 Jmol K Avogadros number NA 6023 10 23 mol 1. Both Avogadros number and the Boltzmann constant were given exact numerical values. Charge of electron e 1602 10 19 C.
The gas constant is also defined as the Avogadro constant N A multiplied by the Boltzmann constant k. When we use gmol we describe the weight of one mole of a given substance. Coulomb constant 14πε 0 8987551792314 10 9 N m 2.
6022 x 10 23 mol-1. The number of units in one mole of any substance is called Avogadros number or Avogadros constant. Electron mass energy MeV m e c 2.
Molar mass is often confused with atomic or molecular mass. Gas Constant in other important equations. Its dimension is work per amount per temperature and the constant is exactly defined as 831446261815324 JK¹mol¹.
Avogadros law Opens a modal The van der Waals equation Opens a modal Partial pressure Opens a modal Practice. 6022 10 23 mol-1. 1602 10-19 C.
The gas constant R is defined as the. Avogadros number is defined to be the number of atoms in 12 grams of carbon and is approximately six followed by 23 zeroes. Avogadros number background radiation 3K bag model quarks ballistic pendulum band theory solids bandwidth Barnards Star barometer barometric formula barrier penetration baryons baseball curve beat frequency Bequerel unit Bernoulli principle beta radioactivity bicycle wheel precession.
Thus the gas constant R can be given as Gas constant R 8314459848 Jmol 1 K 1. Where p is the pressure V is the volume n is the amount of substance of gas R is the universal gas constant and T is the thermodynamic temperature. The constant is also a combination of the constants from Boyles law Charless law Avogadros law and Gay-Lussacs law.
The Avogadro constant is named after the Italian scientist Amedeo Avogadro 17761856 who in 1811 first proposed that the volume of a gas at a given pressure and temperature is proportional to the number of atoms or molecules regardless of the nature of the gas. The Boltzmann constant and the Avogadro constant were similarly determined which separately relate energy to temperature and particle count to amount of substance. Boltzmanns constant Opens a modal Heat capacity at constant volume and pressure Opens a modal Kinetic molecular theory of gases Opens a modal Practice.
The Boltzmann constant is the ratio of the ideal gas constant to the Avogadros constant. The physical significance of k is that it Boltzmann constant symbol k a fundamental constant of physics occurring in nearly every statistical formulation of both classical and quantum physics. Electron charge to mass ratio-em e-17588 x 10 11 C kg-1.
Using equation where m is the mass per molecule of the gas. Faradays Constant F Ne. It is equal to 602214085710 23.
Kinetic molecular theory of gases. Boltzmann constant k 138 10 23 JK. The gas constant corresponds to the work performed by one mole of an ideal gas when heated by 1 K at constant pressure.
0529 x 10-10 m. Avogadros number P pressure default is 1 atmosphere R gas constant entropy due to electronic motion entropy due to rotational motion total entropy entropy due to translation entropy due to vibrational motion T temperature default is 29815 V volume characteristic temperature for rotation in the x y or z plane characteristic temperature for vibration K. The constant is named after Ludwig Boltzmann a 19th-century Austrian physicist who.
Temperature and amount of substance. 138 x 10-23 J K-1.

A Mind Dump Of Mathematics Deriving Boltzmann S Constant

Given Avogadro S Number N 6 02 Xx 10 23 And Boltzmann S Constant K 1 38 Xx 10 23 Youtube

Boltzmann Constant Definition And Units

Mathtype The Gas Constant R Is Defined As The Avogadro Constant Na Multiplied By The Boltzmann Constant K It Is Mostly Known For Appearing In The Ideal Gas Law And Physically

Avogadro S Law And Botlzmann Constant Youtube
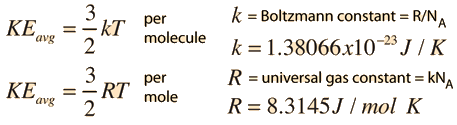
0 Response to "Boltzmann Constant and Avogadro's Number"
Post a Comment